
Many economies are dependent on fish and shellfish and people worldwide rely on food from the ocean as their primary source of protein. Ocean acidification is affecting the entire world’s oceans, including coastal estuaries and waterways. When these organisms are at risk, the entire food web may also be at risk. Certain fish's ability to detect predators is decreased in more acidic waters. It is the conjugate base of the hydrogencarbonate (bicarbonate) 8 ion, HCO 3. It has a molecular mass of 60.01 g/mol and carries a total formal charge of 2. It consists of one carbon atom surrounded by three oxygen atoms, in a trigonal planar arrangement, with D3h molecular symmetry. These changes in ocean chemistry can affect the behavior of non-calcifying organisms as well. Calcium carbonate nucleation driven by ion binding in a biomimetic matrix revealed by In situ electron microscopy, Nature Materials Month Day, Year, DOI: 10.1038/NMAT4193 Journal information. The carbonate ion is the simplest oxocarbon anion. We will need two hydroxide ions to exactly cancel the 2+ charge on Ca 2 +. Calcium ions have a charge of 2+, while nitrate ions have a charge of 1. We will need two potassium ions to balance the charge on the sulfate ion, so the proper chemical formula is K 2 SO 4. Potassium ions have a charge of 1+, while sulfate ions have a charge of 2. From our table, we know that hydroxide has the formula OH and that it carries a 1- charge. the calcium ion and the nitrate ion SOLUTION. Both calcium and carbonate have equal yet opposite charges and hence these ions bind together to form calcium carbonate which is a neutral ionic compound. The carbonate molecule is anionic in nature and has a charge of. 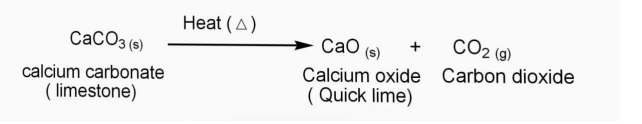 .png)
Decreases in carbonate ions can make building and maintaining shells and other calcium carbonate structures difficult for calcifying organisms such as oysters, clams, sea urchins, shallow water corals, deep sea corals, and calcareous plankton. Calcium is an alkaline earth metalGroup 2 on the periodic tableso it forms ions with a 2+ charge. Thus calcium has a valency of two and its forms a. This increase causes the seawater to become more acidic and causes carbonate ions to be relatively less abundant.Ĭarbonate ions are an important building block of structures such as sea shells and coral skeletons. When CO 2 is absorbed by seawater, a series of chemical reactions occur resulting in the increased concentration of hydrogen ions. The ocean absorbs about 30 percent of the CO 2 that is released in the atmosphere, and as levels of atmospheric CO 2 increase, so do the levels in the ocean. For more than 200 years, or since the industrial revolution, the concentration of carbon dioxide (CO 2) in the atmosphere has increased due to the burning of fossil fuels and land use change.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
